Geostatistical data. Malaria in The Gambia
Paula Moraga
CHICAS, Lancaster University, UK
10 June 2018
In this tutorial we predict malaria prevalence in The Gambia, Africa. We use data of malaria prevalence in children obtained at 65 villages in The Gambia which are contained in the geoR package, and environmental covariates downloaded with the raster package. We show how to obtain malaria prevalence predicitons using the INLA and SPDE approaches, and how to make interactive maps of the prevalence and risk factors using leaflet.
1 Data
First, we load the geoR package and attach the gambia data.
library(geoR)
data(gambia)Next we inspect the data and see it is a data frame with 2035 observations and the following 8 variables:
x: x-coordinate of the village (UTM),y: y-coordinate of the village (UTM),pos: presence (1) or absence (0) of malaria in a blood sample taken from the child,age: age of the child in days,netuse: indicator variable denoting whether (1) or not (0) the child regularly sleeps under a bed-net,treated: indicator variable denoting whether (1) or not (0) the bed-net is treated (coded 0 ifnetuse= 0),green: satellite-derived measure of the greenness of vegetation in the immediate vicinity of the village (arbitrary units),phc: indicator variable denoting the presence (1) or absence (0) of a health center in the village.
dim(gambia)
?gambia
head(gambia)## [1] 2035 82 Data preparation
Data in gambia are given at individual level. Here, we do the analysis at the village level by aggregating the malaria tests by village. We create a data frame called d with columns containing, for each village, the longitude and latitude, the number of malaria tests, the number of positive tests, the prevalence, and the altitude.
2.1 Prevalence
We aggregate the number of tests in each village. We look at the dimension of the unique coordinates and see that the 2035 tests are conducted in 65 unique locations.
dim(gambia)
dim(unique(gambia[, c("x", "y")]))## [1] 2035 8
## [1] 65 2We create a data frame called d containing the village coordinates (x, y), the total number of tests performed (total), the number of positive tests (positive), and the malaria prevalence (prev) in each village. Positive tests results have pos equal to 1 and negative test results have pos equal to 0. Therefore, we can calculate the number of positive tests in each village adding up the elements in gambia$pos. We calculate the prevalence in each village by calculating the proportion of positive tests (number of positive results divided by the total number of tests in each village).
library(dplyr)
d <- group_by(gambia, x, y) %>%
summarize(total = n(),
positive = sum(pos),
prev = positive/total)
head(d)2.2 Transform coordinates
Now, we can plot the malaria prevalence in a leaflet map. leaflet expects data to be specified in geographic coordinates (latitude/longitude). However, the coordinates in the data are in UTM format (Easting/Northing). We transform the UTM coordinates in the data to geographic coordinates using the spTransform() function of the sp package.
First, we create a SpatialPoints object called sps using the sp package where we specify the projection of The Gambia, that is, UTM zone 28. Then, we transform the UTM coordinates in sps to geographic coordinates using spTransform() where we set CRS to CRS("+proj=longlat +datum=WGS84").
library(sp)
library(rgdal)
sps <- SpatialPoints(d[, c("x", "y")], proj4string = CRS("+proj=utm +zone=28"))
spst <- spTransform(sps, CRS("+proj=longlat +datum=WGS84"))Finally, we add the longitude and latitude variables to the data frame d.
d[, c("long", "lat")] <- coordinates(spst)
head(d)2.3 Map prevalence
Now we use leaflet() to construct a map with the locations of the villages and the malaria prevalence. We use addCircles() to put circles on the map, and colour circles according to the value of the prevalences. We choose a palette function with colours from viridis and four equal intervals from values 0 to 1. We use the base map given by providers$CartoDB.Positron so point colours can be distinguised from the background map. We also add a scale bar using addScaleBar().
library(leaflet)
pal <- colorBin("viridis", bins = c(0, 0.25, 0.5, 0.75, 1))
leaflet(d) %>% addProviderTiles(providers$CartoDB.Positron) %>%
addCircles(lng = ~long, lat = ~lat, color = ~pal(prev)) %>%
addLegend("bottomright", pal = pal, values = ~prev, title = "Prevalence") %>%
addScaleBar(position = c("bottomleft"))2.4 Environmental covariates
To model malaria prevalence we will use a covariate that indicates the elevation in The Gambia. This covariate can be obtained with the getData() function of the raster package which can be used to obtain geographic data from anywhere in the world. In order to get the elevation values in The Gambia, we need to call getData() with the three following arguments:
nameof the data set to'alt',countryset to the 3 letters of the International Organization for Standardization (ISO) code of The Gambia (GMB), andmaskset toTRUEso the neighbouring countries are set to NA.
library(raster)
r <- getData(name = 'alt', country = 'GMB', mask = TRUE)We make a map with the elevation raster using the addRasterImage() function of the leaflet package. First we create a palette function pal using the values of the raster (values(r)) and specifying that the NA values are transparent.
pal <- colorNumeric("viridis", values(r), na.color = "transparent")
leaflet() %>% addProviderTiles(providers$CartoDB.Positron) %>%
addRasterImage(r, colors = pal, opacity = 0.5) %>%
addLegend("bottomright", pal = pal, values = values(r), title = "Altitude") %>%
addScaleBar(position = c("bottomleft"))We also add the elevation variable to the data frame d because we will use it as a covariate in the model. To do that we get the altitude values at the villages locations using the extract() function of the raster package. The first argument of this function is the elevation raster (r). The second argument is a two column matrix with the coordinates where we want to know the values, that is, the coordinates of the villages given by d[ ,c("long", "lat")]. We assign the altitude vector to the column alt of the data frame d.
d$alt <- extract(r, d[, c("long", "lat")])2.5 Data
The final dataset is this:
head(d)3 Modelling
In this Section we specify the model to predict malaria prevalence in The Gambia, and detail the steps to fit the model using the INLA and SPDE approaches.
3.1 Model
Conditional on the true prevalence \(P(x_i)\) at location \(x_i\), \(i = 1, \ldots, n\), the number of positive results \(Y_i\) out of \(N_i\) people sampled at \(x_i\) follows a binomial distribution,
\[Y_i|P(x_i) \sim Binomial(N_i, P(x_i)),\]
\[logit(P(x_i)) = \beta_0 + \beta_1 \times \mbox{altitude} + S(x_i)\] Here \(\beta_0\) denotes the intercept, \(\beta_1\) is the coefficient of altitude, and \(S(x_i)\) is a spatially structured random effect which follows a zero-mean Gaussian process with Matern covariance function: \[\mbox{Cov}(S(x_i),S(x_j))=\frac{\sigma^2}{2^{\nu-1}\Gamma(\nu)}(\kappa ||x_i - x_j ||)^\nu K_\nu(\kappa ||x_i - x_j ||).\] Here, \(K_\nu\) is the modified Bessel function of second kind and order \(\nu>0\). \(\nu\) is the smoothness parameter, \(\sigma^2\) denotes the variance, and \(\kappa >0\) is related to the practical range \(\rho = \sqrt{8 \nu}/\kappa\), the distance at which the spatial correlation is close to 0.1.
3.2 Build mesh
Then, we need to build a triangulation mesh that covers The Gambia over which to make the random field discretization. For building the mesh, we use the inla.mesh.2d() function passing the following parameters:
loc: locations coordinates that will be used as initial mesh vertices,max.edge: values denoting the maximum allowed triangle edge lengths in the region and in the extension,cutoff: minimum allowed distance between points.
Here, we call inla.mesh.2d() setting loc equal to the matrix with the coordinates coo. We set max.edge = c(0.1, 5) to use small triangles within the region, and larger triangles in the extension. cutoff = 0.01 to avoid building many small triangles where we have some very close points.
library(INLA)
coo <- cbind(d$long, d$lat)
mesh <- inla.mesh.2d(loc = coo, max.edge = c(0.1, 5), cutoff = 0.01)The number of vertices is given by mesh$n and we can plot the mesh with plot(mesh).
mesh$n
plot(mesh)
points(coo, col = "red")## [1] 686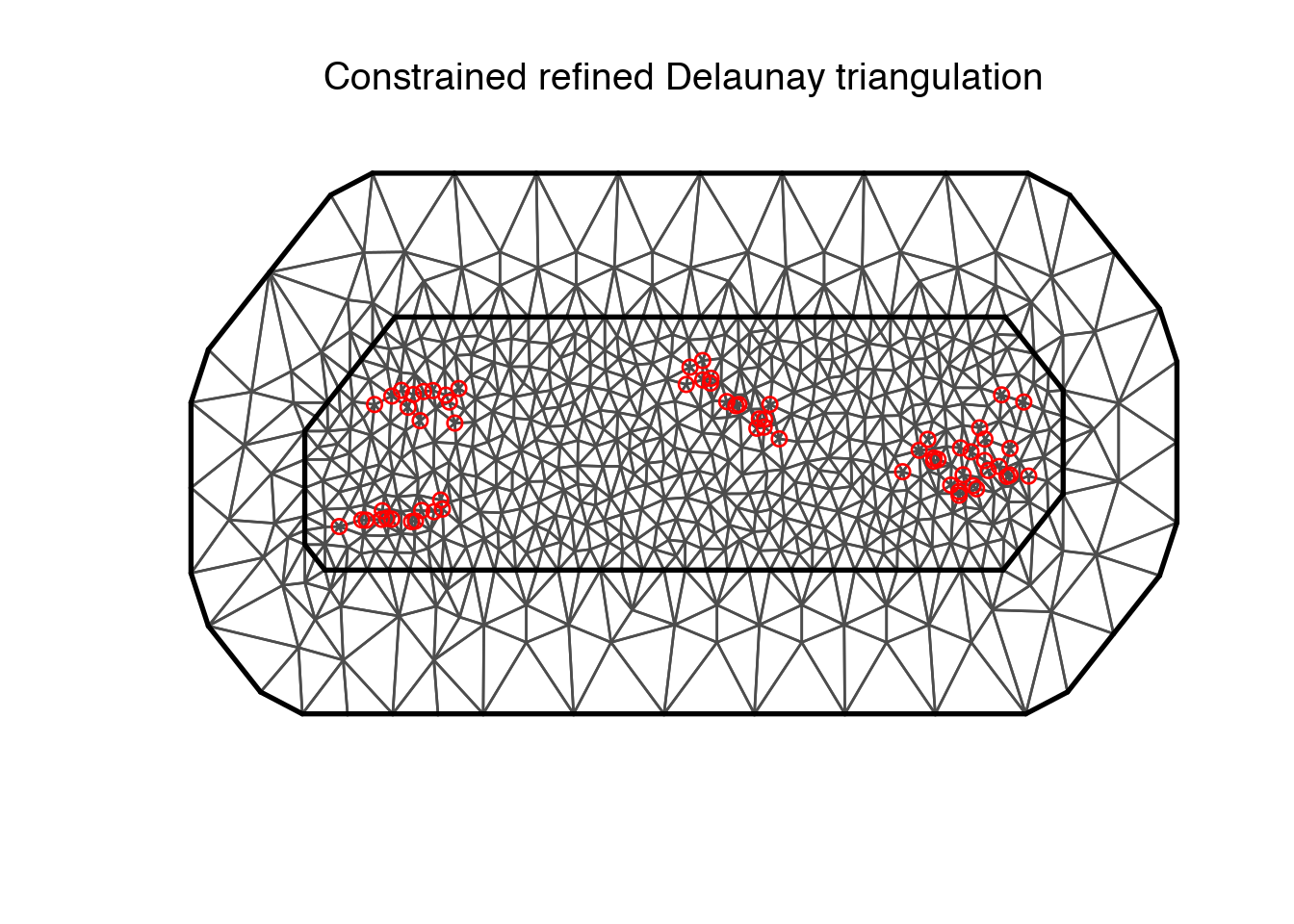
3.3 Build the SPDE model on the mesh
Then, we use the inla.spde2.matern() function to build the SPDE model.
spde <- inla.spde2.matern(mesh = mesh, alpha = 2)Here alpha is a parameter related to the smoothness parameter of the process: \(\alpha=\nu+d/2\). Here, we set the smoothness parameter \(\nu\) equal to 1, and in the spatial case \(d=2\). Therefore alpha=1+2/2=2.
3.4 Index set
Now we generate the index set for the SPDE model. We do this with the function inla.spde.make.index() where we specify the name of the effect (s) and the number of vertices in the SPDE model (spde$n.spde). This creates a list with vector s equal to 1:spde$n.spde, and vectors s.group and s.repl that have all elements equal to 1s and size given by the number of mesh vertices.
indexs <- inla.spde.make.index("s", spde$n.spde)
lengths(indexs)## s s.group s.repl
## 686 686 6863.5 Projector matrix
We need to build a projector matrix A that projects the spatially continuous Gaussian random field at the mesh nodes. The projector matrix A can be built with the inla.spde.make.A() function passing the mesh and the coordinates.
A <- inla.spde.make.A(mesh = mesh, loc = coo)3.6 Prediction data
Here we specify the locations where we want to predict the prevalence. We set the prediction locations to the locations of the the covariate raster data. We can get the coordinates of the raster r with the function rasterToPoints() of the raster package. This function returns a matrix with the coordinates and values of the raster that do not have NA values. We see there are 12964 points.
dp <- rasterToPoints(r)
dim(dp)## [1] 12964 3In this example, we want to use fewer prediction points. We can lower the resolution of the raster by using aggregate() of the raster package. The arguments of the function are
x: the raster object,fact: aggregation factor expressed as number of cells in each direction (horizontally and vertically),fun: function used to aggregate values.
We specify fact = 5 aggregating 5 cells in each direction, and fun = mean to compute the mean of the cell values. We call coop to the matrix of coordinates with the prediction locations.
ra <- aggregate(r, fact = 5, fun = mean)
dp <- rasterToPoints(ra)
dim(dp)
coop <- dp[, c("x", "y")]## [1] 627 33.7 Projector matrix
We also construct the matrix that projects the locations where we will do the predictions.
Ap <- inla.spde.make.A(mesh = mesh, loc = coop)3.8 Stack data for the estimation and prediction
Now we use the inla.stack() function to organize data, effects, and projector matrices. We use the following arguments:
tag: string for identifying the data,data: list of data vectors,A: list of projector matrices,effects: list with fixed and random effects.
We construct a stack called stk.e with data for estimation and we tag it with the string "est". The fixed effects are the intercept (b0) and a covariate (cov). The random effects is the spatial Gaussian Random Field (s). Therefore, in effects we pass a list with a data.frame with the fixed effects, and a list s containing the indices of the SPDE object (indexs). A is set to a list where the second element is A, the projector matrix for the random effects, and the first element is 1 to indicate the fixed effects are mapped one-to-one to the response. In data we specify the response vector and the number of trials. We also construct a stack for prediction that called stk.p. This stack has tag "pred", the response vector is set to NA, and the data is specified at the prediction locations. Finally, we put stk.e and stk.p together in a full stack stk.full.
#stack for estimation stk.e
stk.e <- inla.stack(tag = "est",
data = list(y = d$positive, numtrials = d$total),
A = list(1, A),
effects = list(data.frame(b0 = 1, cov = d$alt), s = indexs))
#stack for prediction stk.p
stk.p <- inla.stack(tag = "pred",
data = list(y = NA, numtrials = NA),
A = list(1, Ap),
effects = list(data.frame(b0 = 1, cov = dp[, 3]), s = indexs))
#stk.full has stk.e and stk.p
stk.full <- inla.stack(stk.e, stk.p)3.9 Model formula
We specify the model formula by including the response in the left-hand side, and the fixed and random effects in the right-hand side. In the formula we remove the intercept (adding 0) and add it as a covariate term (adding b0), so all the covariate terms can be captured in the projector matrix.
formula <- y ~ 0 + b0 + cov + f(s, model = spde)3.10 Call inla()
We fit the model by calling inla() and using the default priors in INLA. We specify the formula, family, data, and options. In control.predictor we set compute = TRUE to compute the posterior means of the predictors. We set link=1 to compute the fitted values (res$summary.fitted.values and res$marginals.fitted.values) with the same link function as the the family specified in the model.
res <- inla(formula, family = "binomial", Ntrials = numtrials,
control.family = list(link = "logit"),
data = inla.stack.data(stk.full),
control.predictor = list(compute = TRUE, link = 1, A = inla.stack.A(stk.full)))3.11 Results
We inspect the results by typing summary(res).
summary(res)##
## Call:
## c("inla(formula = formula, family = \"binomial\", data = inla.stack.data(stk.full), ", " Ntrials = numtrials, control.predictor = list(compute = TRUE, ", " link = 1, A = inla.stack.A(stk.full)), control.family = list(link = \"logit\"))" )
##
## Time used:
## Pre-processing Running inla Post-processing Total
## 2.0369 21.5313 0.3500 23.9182
##
## Fixed effects:
## mean sd 0.025quant 0.5quant 0.975quant mode kld
## b0 -0.1893 0.5271 -1.0806 -0.2377 1.0082 -0.3019 0
## cov -0.0117 0.0107 -0.0325 -0.0118 0.0098 -0.0120 0
##
## Random effects:
## Name Model
## s SPDE2 model
##
## Model hyperparameters:
## mean sd 0.025quant 0.5quant 0.975quant mode
## Theta1 for s -4.021 0.296 -4.598 -4.024 -3.435 -4.031
## Theta2 for s 2.665 0.377 1.915 2.669 3.400 2.681
##
## Expected number of effective parameters(std dev): 43.17(3.967)
## Number of equivalent replicates : 1.506
##
## Marginal log-Likelihood: -216.29
## Posterior marginals for linear predictor and fitted values computed4 Mapping malaria prevalence
Now we map the malaria prevalence predictions in a leaflet map. We can obtain the mean prevalence and lower and upper limits of the 95% credible intervals from res$summary.fitted.values where we need to specify the rows corresponding to the indices of the predictions and the columns "mean", "0.025quant" and "0.975quant". The indices of the stack stk.full that correspond to the predictions are the ones tagged with tag = "pred". We can obtain them by using inla.stack.index() and specifying tag = "pred".
index <- inla.stack.index(stack = stk.full, tag = "pred")$dataprev_mean <- res$summary.fitted.values[index, "mean"]
prev_ll <- res$summary.fitted.values[index, "0.025quant"]
prev_ul <- res$summary.fitted.values[index, "0.975quant"]The predicted values correspond to a set of points. We can create a raster with the predicted values using rasterize() of the raster package. This function is useful for transferring the values of a vector at some locations to raster cells. We transfer the predicted values prev_mean from the locations coop to the raster ra that we used to get the locations for prediction. We use the following arguments:
x = coop, the coordinates where we made the predictions,y = ra, the raster where we transfer the values,field = prev_mean, the values to be transferred (prevalence estimates in locationscoop),fun = mean, this will assign the mean of the values to cells that have more than one point.
r_prev_mean <- rasterize(x = coop, y = ra, field = prev_mean, fun = mean)Now we plot the raster with the prevalence values in a leaflet map. We use a palette function created with colorNumeric(). We could set the domain to the values we are going to plot (values(r_prev_mean)), but we decide to use the interval [0, 1] because we will use the same palette to plot the mean prevalence and the lower and upper limits of the 95% credible interval.
pal <- colorNumeric("viridis", c(0, 1), na.color = "transparent")
leaflet() %>% addProviderTiles(providers$CartoDB.Positron) %>%
addRasterImage(r_prev_mean, colors = pal, opacity = 0.5) %>%
addLegend("bottomright", pal = pal, values = values(r_prev_mean), title = "Prevalence") %>%
addScaleBar(position = c("bottomleft"))We can follow the same approach to create maps with the lower and upper limits of the prevalece estimates. First we create rasters with the lower and upper limits of the prevalences. Then we make the map with the same palette function we used to plot the mean prevalence.
r_prev_ll <- rasterize(x = coop, y = ra, field = prev_ll, fun = mean)
leaflet() %>% addProviderTiles(providers$CartoDB.Positron) %>%
addRasterImage(r_prev_ll, colors = pal, opacity = 0.5) %>%
addLegend("bottomright", pal = pal, values = values(r_prev_ll), title = "LL") %>%
addScaleBar(position = c("bottomleft"))
r_prev_ul <- rasterize(x = coop, y = ra, field = prev_ul, fun = mean)
leaflet() %>% addProviderTiles(providers$CartoDB.Positron) %>%
addRasterImage(r_prev_ul, colors = pal, opacity = 0.5) %>%
addLegend("bottomright", pal = pal, values = values(r_prev_ul), title = "UL") %>%
addScaleBar(position = c("bottomleft"))

This work by Paula Moraga is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License.
5 Session Info
- This document has been created in RStudio using R Markdown and related packages.
- For R Markdown, see http://rmarkdown.rstudio.com
- For details about the OS, packages and versions, see detailed information below:
sessionInfo()## R version 3.5.0 (2018-04-23)
## Platform: x86_64-apple-darwin15.6.0 (64-bit)
## Running under: macOS High Sierra 10.13.4
##
## Matrix products: default
## BLAS: /Library/Frameworks/R.framework/Versions/3.5/Resources/lib/libRblas.0.dylib
## LAPACK: /Library/Frameworks/R.framework/Versions/3.5/Resources/lib/libRlapack.dylib
##
## locale:
## [1] C
##
## attached base packages:
## [1] stats graphics grDevices utils datasets methods base
##
## other attached packages:
## [1] INLA_17.06.20 Matrix_1.2-14 raster_2.6-7 leaflet_2.0.1
## [5] rgdal_1.3-2 sp_1.3-1 bindrcpp_0.2.2 geoR_1.7-5.2
## [9] forcats_0.3.0 stringr_1.3.1 dplyr_0.7.5 purrr_0.2.5
## [13] readr_1.1.1 tidyr_0.8.1 tibble_1.4.2 ggplot2_2.2.1
## [17] tidyverse_1.2.1 captioner_2.2.3 bookdown_0.7 knitr_1.20
##
## loaded via a namespace (and not attached):
## [1] viridis_0.5.1 httr_1.3.1
## [3] splines_3.5.0 jsonlite_1.5
## [5] viridisLite_0.3.0 modelr_0.1.2
## [7] shiny_1.1.0 assertthat_0.2.0
## [9] cellranger_1.1.0 yaml_2.1.19
## [11] pillar_1.2.3 backports_1.1.2
## [13] lattice_0.20-35 glue_1.2.0
## [15] digest_0.6.15 RColorBrewer_1.1-2
## [17] promises_1.0.1 rvest_0.3.2
## [19] colorspace_1.3-2 htmltools_0.3.6
## [21] httpuv_1.4.3 plyr_1.8.4
## [23] psych_1.8.4 pkgconfig_2.0.1
## [25] broom_0.4.4 haven_1.1.1
## [27] xtable_1.8-2 scales_0.5.0
## [29] later_0.7.3 MatrixModels_0.4-1
## [31] lazyeval_0.2.1 cli_1.0.0
## [33] mnormt_1.5-5 magrittr_1.5
## [35] crayon_1.3.4 readxl_1.1.0
## [37] mime_0.5 evaluate_0.10.1
## [39] nlme_3.1-137 MASS_7.3-50
## [41] xml2_1.2.0 foreign_0.8-70
## [43] RandomFieldsUtils_0.3.25 Cairo_1.5-9
## [45] tools_3.5.0 hms_0.4.2
## [47] munsell_0.5.0 compiler_3.5.0
## [49] rlang_0.2.1 grid_3.5.0
## [51] RandomFields_3.1.50 rstudioapi_0.7
## [53] htmlwidgets_1.2 crosstalk_1.0.0
## [55] base64enc_0.1-3 tcltk_3.5.0
## [57] rmarkdown_1.10 gtable_0.2.0
## [59] codetools_0.2-15 reshape2_1.4.3
## [61] R6_2.2.2 splancs_2.01-40
## [63] gridExtra_2.3 lubridate_1.7.4
## [65] bindr_0.1.1 rprojroot_1.3-2
## [67] stringi_1.2.3 parallel_3.5.0
## [69] Rcpp_0.12.17 png_0.1-7
## [71] tidyselect_0.2.4 xfun_0.1Page built on: 16-Jun-2018 at 10:24:49.
2018 | SciLifeLab > NBIS > RaukR